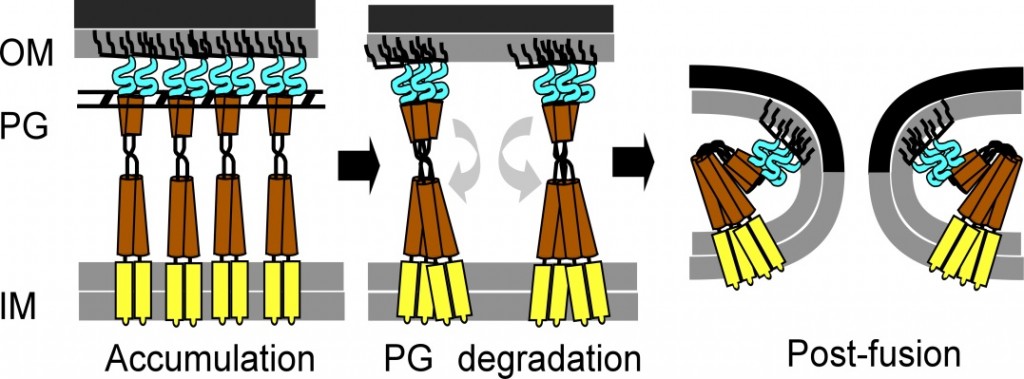
Spanins are essential lysis proteins of bacteriophages (phages) that infect Gram-negative bacteria. In the last step of the lysis event that terminates the infection cycle and liberates the progeny virions, Rz (yellow and brown in figure below) and Rz1 (blue), the spanin subunits of phage lambda, form a complex that disrupts the outer membrane of lambda’s host, E. coli.
Our model for spanin function features lysis of the host cell by fusion of the inner and outer membrane (see figure above). This scheme is analogous to the membrane fusion events in human Golgi trafficking, neurotransmitter release, viral entry, and other important procesess. Thus this system is potentially a valuable model system for the membrane fusion in general. However mechanistic details of spanin function are just now being elucidated. One method to probe the function of spanins is to alter amino acid(s) of Rz or Rz1 and evaluate the phenotype of the mutant spanin complex. We are interested in finding mutants that prevent lysis in vivo and block function in our membrane fusion assay. Using mutagenesis screens and selections, we hope to discover residues that play in important role in spanin function.
An undergraduate research apprentice will have the opportunity to apply and refine microbiological techniques, while simultaneously using biochemical techniques such as PCR, DNA extraction, and mutagenesis.
Contact Dr. Young to get more information or apply.